Kyu-Hwa Hur, M.D.
Department of Laboratory Medicine
Why the HYDRASHIFT assay is needed in the era of therapeutic monoclonal antibodies
Treatment of multiple myeloma has advanced
remarkably over the past several years. As monoclonal antibodies have become
established as standard therapy, patient survival rates and treatment responses
have significantly improved. In particular, anti-CD38 monoclonal antibodies,
including daratumumab, are widely used from initial treatment to
relapsed/refractory patients and have become a central pillar in multiple
myeloma therapy.
Ref) Cancer Manag Res. 2020;12:7891-7903.
Fig. 1. Daratumumab mechanism of action.
However, advances in treatment strategies have introduced new challenges in the testing environment. Protein electrophoresis and immunofixation electrophoresis remain essential tests for the diagnosis of multiple myeloma and for evaluating treatment response. However, in situations where therapeutic monoclonal antibodies are present in the body, the therapeutic antibody itself is observed in protein electrophoresis and immunofixation electrophoresis in the same form as monoclonal proteins that have abnormally proliferated, thereby causing confusion in the interpretation of test results.
Daratumumab is a humanized monoclonal antibody with an IgG-κ structure. Due to this structural characteristic, it can form a monoclonal band at nearly the same position as the patient’s IgG-κ type M-protein in immunofixation electrophoresis. The problem is that, based on the test results alone, it is not possible to distinguish whether this band represents a monoclonal protein derived from tumor cells or a residual monoclonal antibody , daratumumab, administered for treatment purposes.
A situation frequently observed in clinical practice is that, although the monoclonal band has disappeared or is barely detectable in protein electrophoresis, an IgG-κ band is still observed in immunofixation electrophoresis. As a result, even in a state of complete response, the test may still be interpreted as positive on immunofixation electrophoresis, leading to failure to meet the criteria for complete response. Conversely, there is also the possibility that a small amount of monoclonal protein may be masked by the residual therapeutic monoclonal antibody, resulting in underestimation.
In other words, when a monoclonal band is observed in the test results, it is difficult to clearly distinguish whether it originates from a monoclonal protein produced by the patient’s tumor cells or from a monoclonal antibody administered for treatment. This interpretive ambiguity extends beyond a simple issue of uncertainty in laboratory test result reporting and directly affects the patient’s treatment course and overall prognosis, including decreased accuracy in evaluating treatment response, delays in decisions regarding treatment discontinuation or maintenance, and uncertainty in establishing follow-up testing strategies.
The HYDRASHIFT assay was developed specifically to resolve these issues in test interpretation and to improve the reliability of immunofixation electrophoresis results.
What is the HYDRASHIFT assay?
The
HYDRASHIFT assay is an advanced test designed to remove or separate
interference caused by therapeutic monoclonal antibodies during the process of
immunofixation electrophoresis performed in patients with multiple myeloma,
thereby enabling more accurate evaluation of monoclonal proteins derived from
multiple myeloma. The HYDRASHIFT daratumumab assay was designed to identify and
separate interference caused by daratumumab administration and is a commercially
available assay approved by the Ministry of Food and Drug Safety that is
already widely used worldwide.
The principle of the HYDRASHIFT assay involves the addition of an anti-daratumumab antibody that specifically binds to daratumumab present in serum, thereby altering the electrophoretic migration pattern of the daratumumab monoclonal antibody. In this process, the daratumumab monoclonal antibody migrates away from the original IgG-κ region to a different location or is observed in a transformed complex form. As a result, in immunofixation electrophoresis, the band caused by the therapeutic monoclonal antibody and the band caused by monoclonal protein derived from tumor cells can be visually distinguished. Thus, the HYDRASHIFT assay can be understood as a method that enables more accurate interpretation of conventional immunofixation electrophoresis results.
HYDRASHIFT Assay Procedure
The core principle of the HYDRASHIFT assay is to use
an antibody that specifically binds to the therapeutic monoclonal antibody, thereby
altering the electrophoretic migration pattern of the therapeutic antibody.
Through this process, it enables visual differentiation of whether the band
observed in immunofixation electrophoresis is caused by a therapeutic monoclonal
antibody or by a monoclonal protein.
The test procedure is basically similar to that of conventional IFE, with the addition of a processing step using HYDRASHIFT reagent. The general procedure can be summarized as follows.


Fig. 2. False-positive immunofixation electrophoresis results caused by a therapeutic monoclonal antibody
(before and after treatment with the HYDRASHIFT daratumumab reagent)
(A) Before reagent treatment, (B) After reagent treatment. After reagent treatment, it can be confirmed that the therapeutic
monoclonal antibody band has migrated to the alpha1 globulin zone. The final result is a pseudopositive monoclonal band
caused by the daratumumab monoclonal antibody.
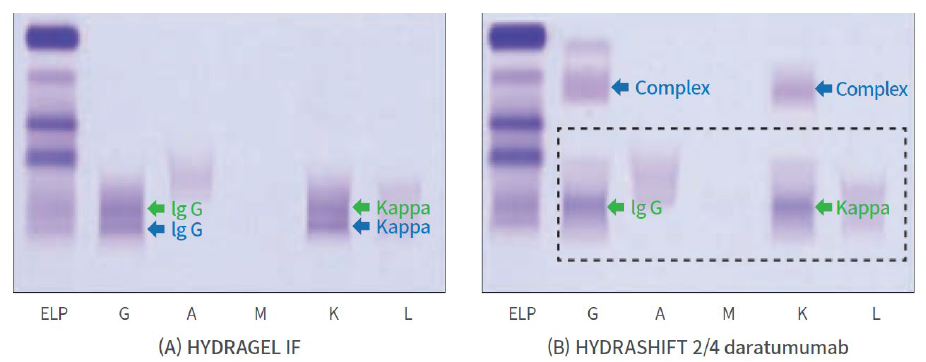
Fig. 3. Positive immunofixation electrophoresis results due to residual monoclonal protein in a patient
(before and after treatment with the HYDRASHIFT daratumumab reagent)
(A) Before reagent treatment, (B) After reagent treatment. The green monoclonal band represents residual monoclonal protein,
and the blue monoclonal band represents the therapeutic monoclonal antibody. After reagent treatment, it can be confirmed
that the two monoclonal bands are clearly distinguished.
Clinical Significance of the Assay
The HYDRASHIFT
assay has the advantage of high specificity. The anti-daratumumab
antibody is designed to bind specifically to daratumumab, and therefore does
not affect other immunoglobulins present in the patient’s serum or monoclonal
proteins derived from multiple myeloma. Accordingly, unnecessary confusion in the
interpretation of test results can be minimized.
From a laboratory perspective, it can greatly improve the objectivity and reliability of test result interpretation. In conventional immunofixation electrophoresis, when an IgG-κ monoclonal band is observed, it is not possible to exclude the possibility of interference by therapeutic antibodies in patients treated with daratumumab, and thus the test results inevitably remain subject to interpretation based on estimation. This has become a rapidly emerging issue in recent years as monoclonal antibodies have been established as standard therapy for multiple myeloma. In line with changes in standard treatment trends, the application of the HYDRASHIFT assay enables direct confirmation of interference through objective and visual migration of the monoclonal band, rather than relying on estimation.
From a clinical perspective, treatment response can be evaluated more accurately. In the treatment of hematologic malignancies, including multiple myeloma, the assessment of treatment response is a key criterion for determining subsequent treatment strategies and prognosis. If it is possible to clearly distinguish whether a positive monoclonal band observed in immunofixation electrophoresis is due to residual monoclonal protein or a therapeutic antibody through the HYDRASHIFT assay, the reliability of treatment response assessment can be greatly improved. In addition, unnecessary prolongation of treatment or additional testing can be significantly reduced. If monoclonal bands caused by therapeutic antibodies are misinterpreted as residual disease positivity, there is a risk of unnecessary continuation of treatment or performance of additional invasive tests.
The HYDRASHIFT assay is not merely an additional test, but plays a role in making both laboratory and clinical workflows and decision-making processes more clear and safer.
References

Test Information
GC Labs code | Test item | Specimen (mL) | Test schedule/ | Test method |
F490 | Daratumumab-specific protein [IFE] | Serum 1.0 | Tue / 2 days | IFE |
* The above information is as April 1, 2026, and may change later. Please visit our website for the latest updates

